Abstract
Recipient chimerism increase has been used to predict leukemia relapse in post-hematopoietic cell transplant (HCT) patients with conventional GVHD prophylaxis. However, the value of recipient chimerism increase in patients with post-transplant cyclophosphamide (PTCy) is not clear. We compared PTCy to conventional GVHD prophylaxis (non-PTCy) patients regarding engraftment kinetics and the clinical significance of the 2 chimerism parameters, increasing mixed chimerism (IMC) and degree of recipient chimerism increase at the first event (Δ increase). We studied both total and T-cell-specific chimerism. While leukemia relapse is manifested by an increase in total cell recipient chimerism, an increase in T-cell-specific recipient chimerism may be more impactful in predicting relapse because of the effect of increased T-cell-specific chimerism on the graft-versus-leukemia effect.
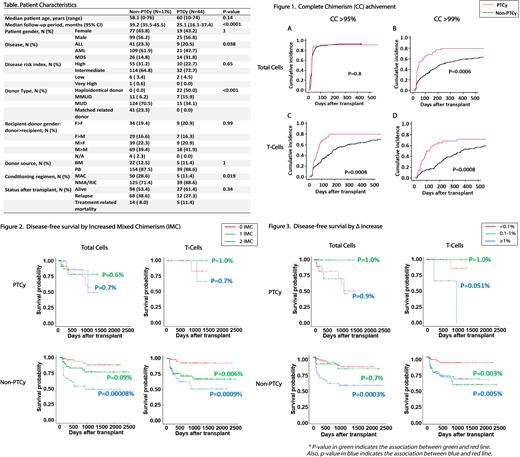
A total of 220 patients (PTCy: 44, non-PTCy: 176) with AML, MDS, and ALL underwent HCT at our institution from January 2014 to September 2020 and were included in this study (Table). Chimerism was tested at least monthly for the first 3 months, followed by every 3 months up until 1-year post-HCT, and then every 6-12 months thereafter. Short tandem repeat or quantitative PCR were used when percent recipient chimerism was ≥5% and <5%, respectively. Cumulative incidence of competing events and Gray's test were applied for engraftment analysis. Relapse and non-relapse mortality were considered as competing risks for engraftment. Mantel-Byar test and Simon-Makuch plot with landmark analysis were used to visualize disease-free survival (DFS) curves. The Cox proportional hazards model with time-dependent covariates was performed to identify the factors affecting relapse.
PTCy patients achieved complete donor chimerism (CC) in total cells earlier at a deeper level (>99%) as compared to non-PTCy patients. Deeper total cell CC (>99%) was achieved in 79.5% of PTCy vs. 51% of non-PTCy patients at day 250, while CC (>95%) was achieved in almost 90% of patients in both groups within 100 days (Figure 1A and B). In comparison, the percentage of PTCy patients achieving T-cell-specific CC was significantly higher at day 250 post-HCT: CC (>95%/>99%) was 79.7%/68.4% in PTCy patients vs. 56.1%/37.5% in non-PTCy patients (Figure 1C and D).
To evaluate their impact in predicting relapse, IMC was stratified into no IMC, 1 IMC (≥1 nonconsecutive IMC), and 2 IMC (≥2 consecutive IMC), and degree of recipient chimerism increase at the first event (Δ increase) was stratified into <0.1% (no Δ increase), 0.1-1%, and ≥1%.
Two IMC (total), 1 IMC (T-cell), and 2 IMC (T-cell) groups were associated with shorter DFS in non-PTCy patients but not in PTCy patients (Figure 2). One and 2 IMC groups (both total and T-cell) were associated with relapse risk in non-PTCy patients. Furthermore, 1 IMC (T-cell) in non-PTCy patients showed a strong association in relapse risk (HR 7.0 (95%CI 2.83-17.8) p<0.0001).
Δ increase ≥1% (total and T-cell) and Δ increase ≥0.1% (T-cell) were associated with shorter DFS in non-PTCy patients, while only Δ increase ≥1% (T-cell) only showed a trend towards shorter DFS in PTCy patients (Figure 3). The Cox regression model showed Δ increase ≥1% in both total, and T-cell chimerism was associated with relapse risk in non-PTCy patients (HR 6.4 (95%CI 2.9-14.2) p<0.0001 and HR 7.2 (95%CI 2.9-18.1) p<0.0001, respectively). Δ increase ≥0.1% (T-cell) in non-PTCy patients was also associated with relapse risk (HR 7.2: 95%CI 2.5-20.4, p<0.0001). In comparison, no association was found between Δ increase and relapse risk in PTCy patients.
This is one of the most extensive studies investigating engraftment kinetics and the association of total and T-cell recipient chimerism increase to predict leukemia relapse in PTCy and non-PTCy HCT recipients. We found that PTCy HCT recipients achieved deeper engraftment earlier as compared to non-PTCy recipients. In addition, the two chimerism parameters (IMC and Δ increase) are less reliable in predicting relapse in PTCy than non-PTCy recipients. However, other factors, such as disease type, conditioning regimen, and donor HLA disparity, may have affected engraftment kinetics and the significance of chimerism parameters. Further investigations are warranted to elucidate the impact of the engraftment kinetics and recipient chimerism increase to predict relapse, especially in the PTCy setting.
Rakszawski: SeaGen: Speakers Bureau. Naik: Takeda: Other: Virtual Advisory Board Member ; Sanofi: Other: Virtual Advisory Board Member ; Kite: Other: Virtual Advisory Board Member. Rybka: Spark Therapeutics: Consultancy; Merck: Consultancy. Claxton: Astellas: Other: Clinical Trial; Novartis: Research Funding; Astex: Research Funding; Cyclacel: Research Funding; Daiichi Sankyo: Research Funding; Incyte: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal